
Shared Concern for Health - Worldwide
Helix quality and new product formulation has provided us impetus in our endeavor outside Pakistan. Now in our selected market, Helix is considered as one of the major player from Pakistan.
Product Range for Global Markets
Helix Pharma offers a diverse and expanding product portfolio covering:
Prescription Pharmaceuticals (oral solids, liquids, injectable, Ophthalmic/otic)
OTC & Nutraceuticals (Oral solid and sachet)
All our formulations are developed with a focus on quality, efficacy, and affordability, addressing regional therapeutic needs and public health priorities.

Strategic Partnerships
We welcome global collaborations in:
01
Product Licensing and Co-Marketing
02
Contract Manufacturing
03
Distribution and Supply Partnerships
04
Technology Transfer and Brand Representation
Through these alliances, Helix Pharma aims to deliver trusted healthcare solutions to new markets while fostering long-term relationships built on mutual growth and integrity.

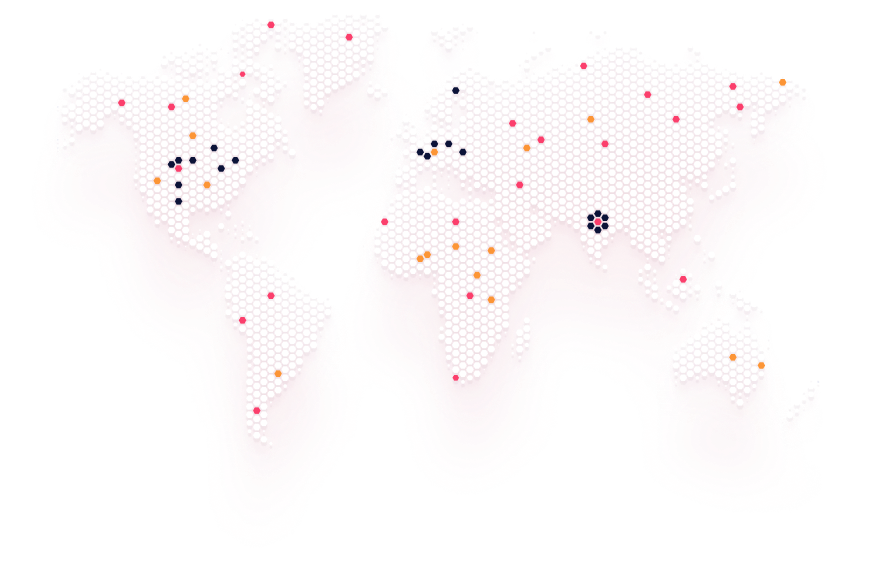
Our Global Presence
With a growing footprint across Asia, the Middle East, Africa, and the CIS region, Helix Pharma continues to strengthen its global presence through exclusive partnerships, strategic licensing, and distributor alliances.
We collaborate with trusted partners to make our products accessible in compliance with each country’s regulatory standards.
Regions of Operation
We welcome global collaborations in :
C.I.S ( Azerbaujan, Monogolia, Turkmenistan, Uzbekistan )
Middle East (Iraq, Yemen)
Africa (Lvory Coast, Kenya, Lesotho, Malawi, Mozambique, Namibia, Rwanda, Somalia, Uganda, zambia )
South and Far East Asia (Sri Lanka, Cambodia, Myanmar, Philippines)
Commitment to Compliance & Quality
Every product we export is backed by comprehensive regulatory documentation and quality assurance systems aligned with DRAP, WHO and PICs requirements.
We ensure compliance at every stage — from dossier submission to final shipment — maintaining our reputation as a dependable global healthcare partner.

Export Capabilities
Our state-of-the-art manufacturing facilities are certified to meet cGMP, ISO, and WHO standards, enabling us to supply products globally with confidence.
We handle end-to-end export operations including:
Regulatory dossier submissions & export documentation
Product registration support
Customization of packaging and labeling for each market
Temperature-controlled logistics and GDP-compliant shipment
International Regulatory Body Certifications
Ministry of Heath – Cambodia
National Medicine Regulatory Authority – Sri Lanka
Pharmaceutical Regulations – Ivory Coast
Pharmacy and Poisons Board – Kenya
Food and Drug Administration – Philippines
Ministry of Health – Senegal
Tanzania Food and Drug Authority (in-process)
National Drug Authority – Uganda (in-process)

Our Locations
Ireland
Kenya
Sri lanka
Cambodia
Uganda
